
PDF) Dissolution testing of bilayer tablets: Method development, validation and application in post-marketing quality evaluation

PDF) Development and Validation of Dissolution Tests for Fexofenadine Hydrochloride Capsules and Coated Tablets | Ana Rita Paim - Academia.edu

PDF) Development and Assessment of a USP Apparatus 3 Dissolution Test Method for Sustained-Release Nevirapine Matrix Tablets
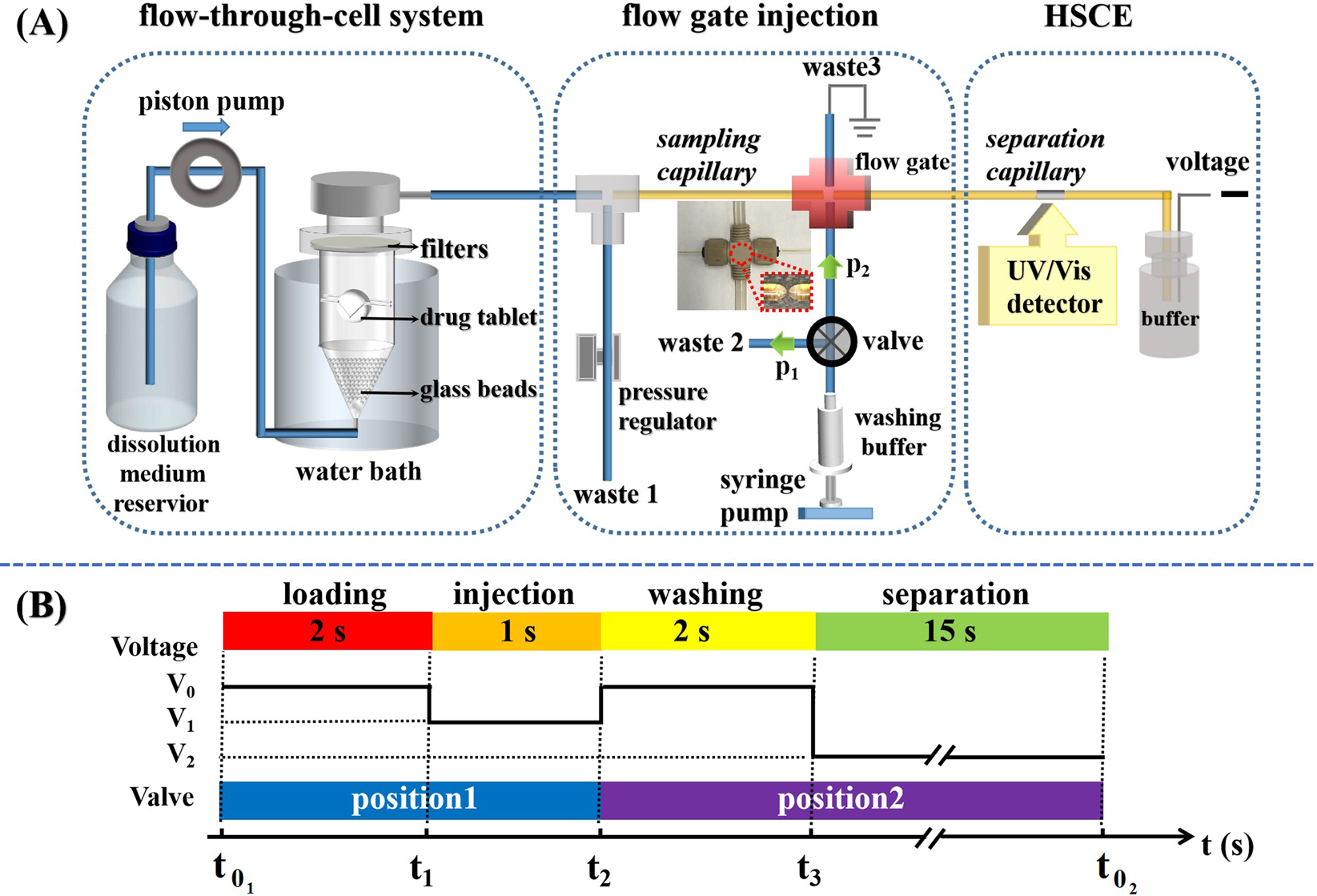
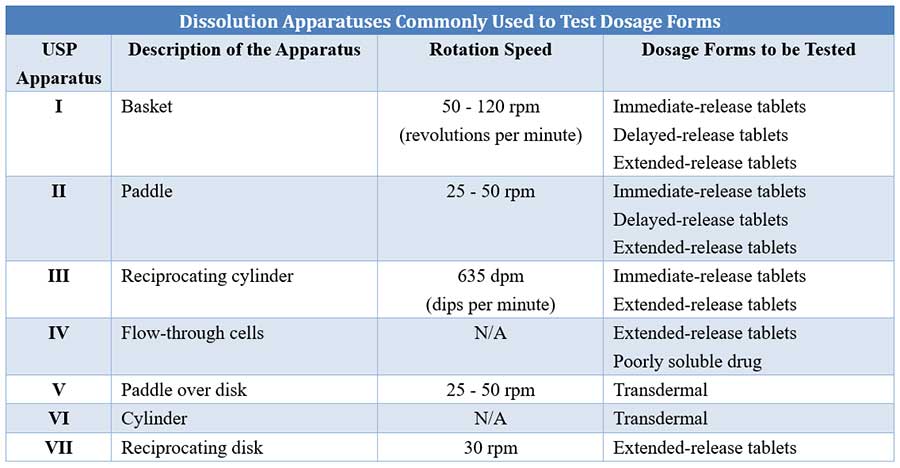
Automatic Dissolution Testing with High-Temporal Resolution for Both Immediate-Release and Fixed-Combination Drug Tablets | Scientific Reports
![PDF] Examination of Selection of Immediate Release Dissolution Acceptance Criteria | Semantic Scholar PDF] Examination of Selection of Immediate Release Dissolution Acceptance Criteria | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/4083b512d5f994055c08b85a648ef1f31b2e9534/1-Table1-1.png)








![PDF] The Significance of disintegration testing in pharmaceutical development | Semantic Scholar PDF] The Significance of disintegration testing in pharmaceutical development | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/92a193f174837cd54c0ca8eb588ad0f3e936abc1/3-Table2-1.png)

![PDF] History and Evolution of the Dissolution Test | Semantic Scholar PDF] History and Evolution of the Dissolution Test | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/e445cfd8d997cbb7a15cc00e6cf58d6cb6415222/5-Figure2-1.png)









