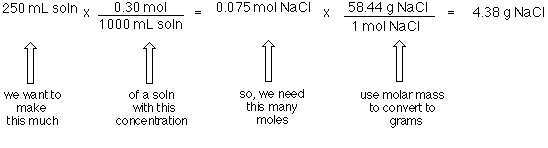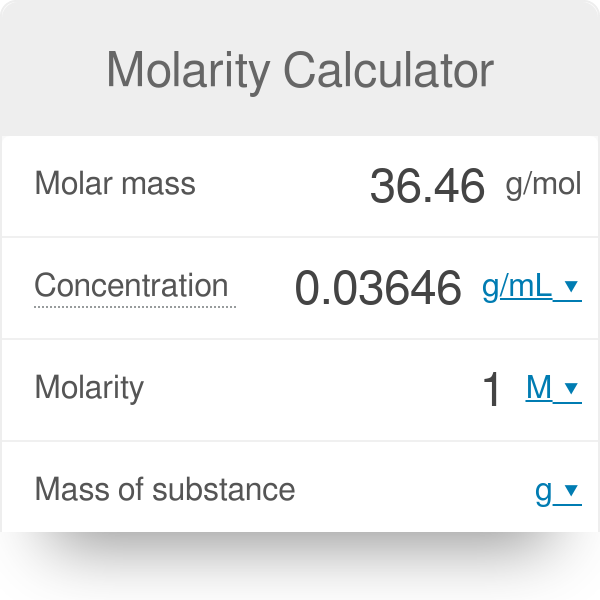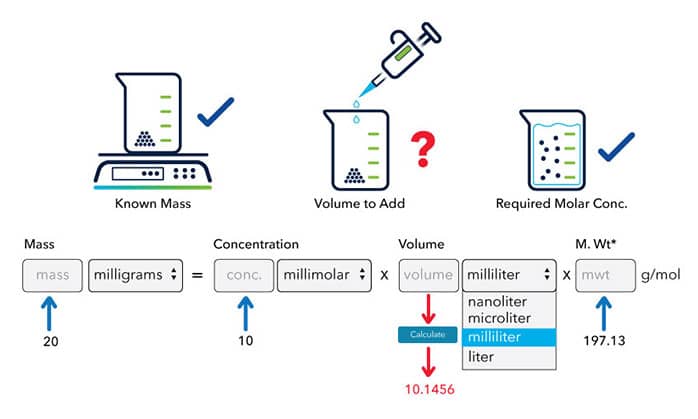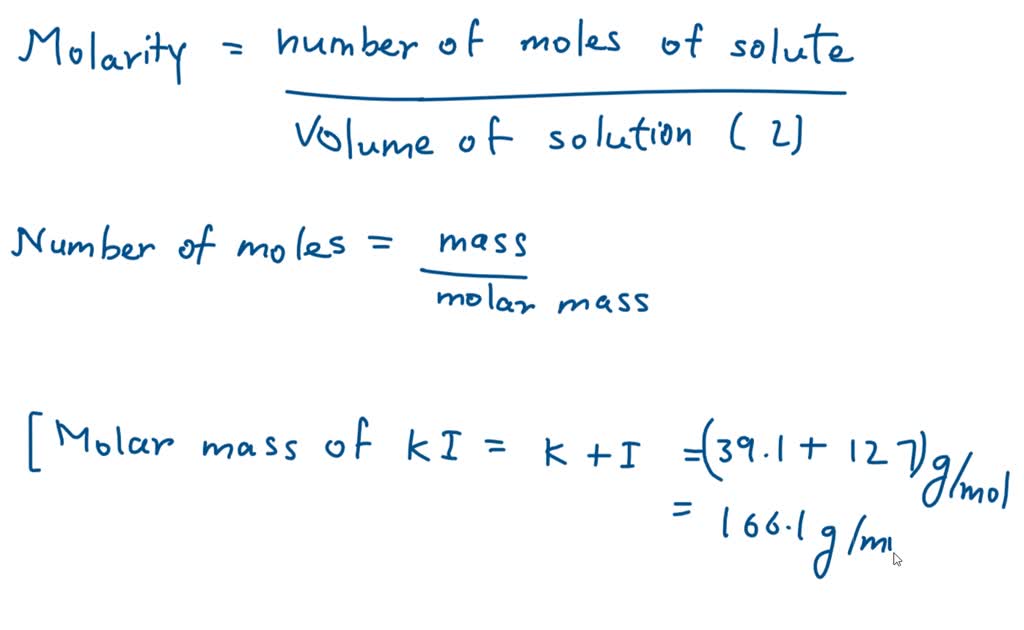
SOLVED: Calculate the molarity of each solution. 33.2 mg of KI in 118 mL of solution Express your answer using three significant figures.

Molarity Dilution Problems Solution Stoichiometry Grams, Moles, Liters Volume Calculations Chemistry - YouTube

I have H2O2 of molecular wt 34.01gm and 30% w/v. What does it mean that I am not getting it and I want to prepare 0.1M solution, how can i? | ResearchGate
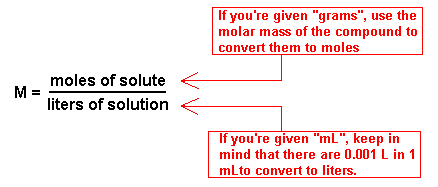
What is the molarity of a solution that contains 50.0 g of Mg(NO_3)_2 per 225 mL of solution? | Socratic

CALCULATIONS I. Reconstituting Cytokines/Growth Factors Need To Supplement Cultures With Recombinant Growth factors/Cytokines Issues To Consider –Recombinant. - ppt download
![SOLVED: CHEM 121: Introduction to Chemistry Concentration is a conversion factor: Molarity [M, moles solute/L solution] lets you convert between volume of solution and moles of solute. Dilution Equation: M1V1 = M2V2. SOLVED: CHEM 121: Introduction to Chemistry Concentration is a conversion factor: Molarity [M, moles solute/L solution] lets you convert between volume of solution and moles of solute. Dilution Equation: M1V1 = M2V2.](https://cdn.numerade.com/ask_images/94726641ed1f4ed69c39f39139455c2d.jpg)