
SOLVED: Calculate the amount of heat (in joules) required to convert 292.5 g of water at 20.0°C to steam at 130.0°C. (Sp. heat of H2O(l) = 4.18 J/g-°C, Sp. heat of H2O(g) =

Calculate the heat required to convert 3 kg of ice -12°C kept in a calorimeter to steam 100°C atmospheric pressure. Given specific heat capacity ice = 2100 J kg '°C !, specific

nus. TI I J The amount of heat energy required to convert 1 kg of ice -10°C to water 100°C is 7,77,000 J. Calculate the specific latent heat of ice. Specific heat

SOLVED: Calculate the total heat, in joules, needed to convert 15.0 g of liquid ethanol at 25.0 °C to gas at its boiling point of 78.0 °C. Ethanol has a specific heat

Work done in converting one gram of ice at 10∘ C into steam at 100∘ C is [Given, specific heat of ice c i =0.5 cal g 1∘ C 1, specific heat






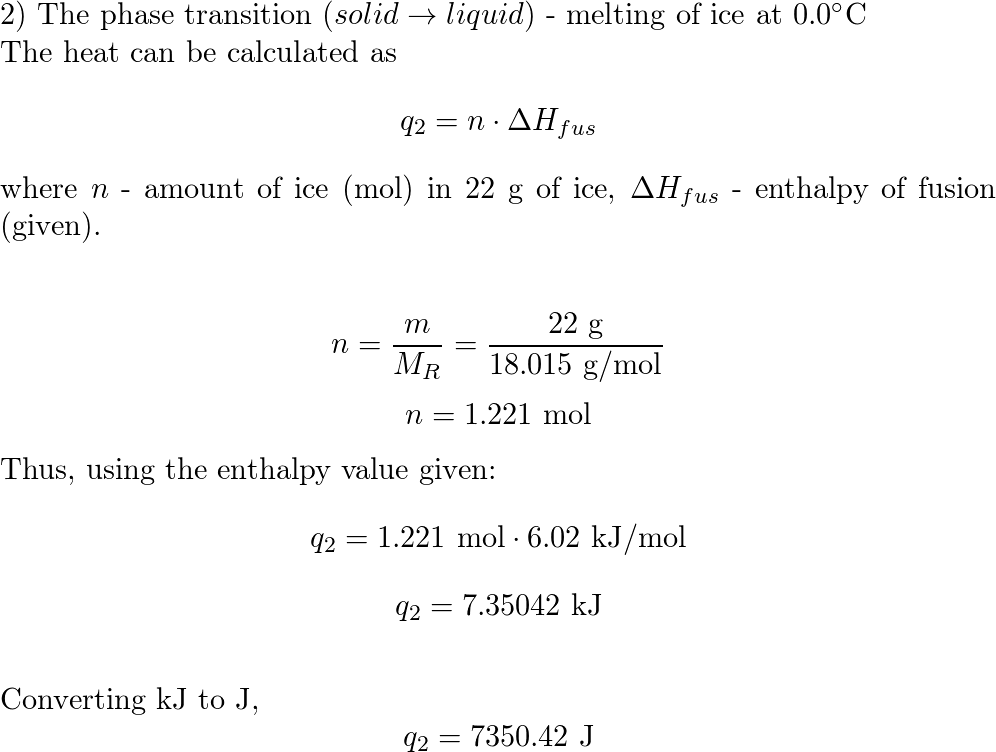



![ANSWERED] What amount of heat (in kJ) is required t... - Physical Chemistry - Kunduz ANSWERED] What amount of heat (in kJ) is required t... - Physical Chemistry - Kunduz](https://media.kunduz.com/media/sug-question/raw/54105585-1657456603.3299727.jpeg)



![Tamil] Convert 1 kWh in joule. Tamil] Convert 1 kWh in joule.](https://static.doubtnut.com/ss/web-overlay-thumb/2401537.webp)

